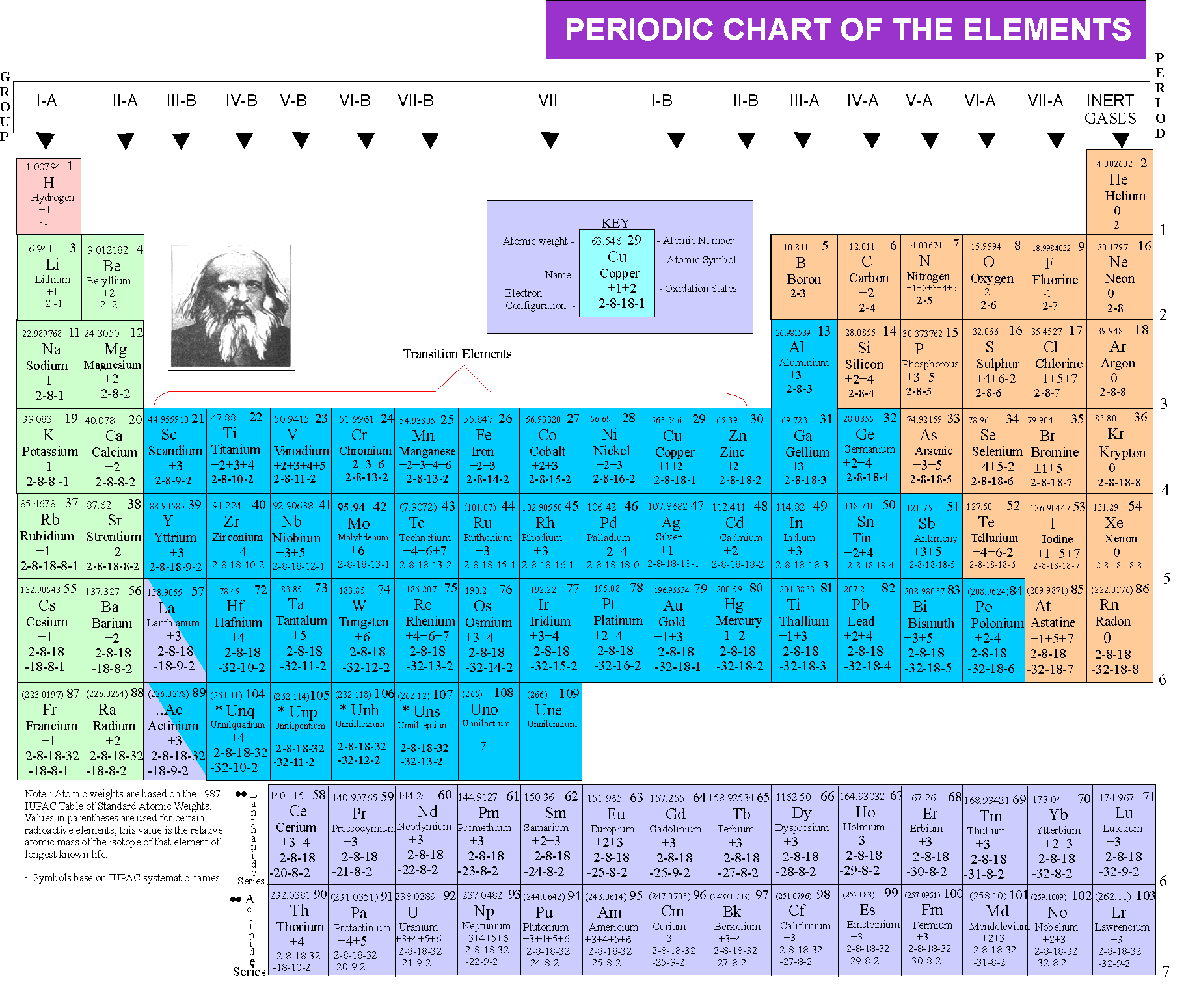
Like the other alkaline earth metals (group 2 of the periodic table) it occurs naturally only in combination with other elements and it almost always has an oxidation state of +2. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Therefore, the empirical formula of aluminium oxide is Al 2O 3. Magnesium is a chemical element with the symbol Mg and atomic number 12. Each oxide has a charge of 2-, which means that we can get 6 negative charges by having 3 oxides. To balance the charges you would need 2 chloride ions for every magnesium ion: 2 negative charges. Using a simple, general trend for the ionic charge for elements on the Periodic Table, in this video we find the ionic charge for Magnesium (Mg). Each aluminium has a charge of 3+, which means that we can get 6 positive charges by having 2 aluminiums. The magnesium chloride has to be electrically neutral overall. In contrast, calcium has 18 inner shell electrons that shield the. This means that we can balance the charge of this ionic compound by having 6 positive charges and 6 negative charges. However, magnesium has 10 inner shell electrons that shield the charge of 10 of these protons. The first number that 3 and 2 go into is 6 (the LCM is 6). The balancing for aluminium oxide is slightly different because the positive and negative charges do not go into each other the 3 positive charge from aluminium and the 2 negative charge from oxygen do not go into each other. whereas magnesium ions (Mg2+) is essential for certain enzymes, muscles. 
1 Comparison of 3d (gray) and 4s (red) electron clouds for a vanadium atom. While these are the most common valences, the real behavior of electrons is less simple. It is the very strong attractive force of this small fraction of the total 4 s electron density that lowers the energy of the 4 s electron below that of the 3 d. After the electron configuration, the last shell of the magnesium atom has two electrons. You may assume that the valences of the elementsthe number of electrons with which an atom will bond or formare those that can be derived by looking at the groups (columns) of the periodic table. The overall charge for an ionic compound must be 0, which means that the charges from aluminium and oxygen must balance out. Trends in Ionic Charge: Meaning Periodic Table Ionisation Energy. The ground state electron configuration of magnesium is 1s 2 2s 2 2p 6 3s 2. Periodic Table With Charges Alkali Metal Alkaline Earth Transition Metal Basic Metal Metalloid Nonmetal Halogen Noble Gas Lanthanide H Hydrogen 1.008 Li Lithium 6.941 Be Beryllium 9.012 Mg Magnesium 24.305 K Potassium 39.098 Ca Calcium 40.078 Lanthanoids Actinoids Kr Krypton 83. Chemical element, metallic, symbol Mg, situated in group IIa in the periodic table, atomic number: 12, atomic weight: 24,312. Oxygen is in group 6 in the periodic table, which means that it will gain 2 electrons resulting in it having a 2 negative charge (O 2-). Aluminium is a metal and is in group 3 in the periodic table, which means that it will lose 3 electrons resulting in it having a 3 positive charge (Al 3+). What is the empirical formula of aluminium oxide?Īluminium oxide is made out of aluminium and oxygen atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
